AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Calculating absolute molar entropy of f24/23/2024  The standard entropies of formation, S ° (J/K) for the reactants and products are: H 2S( g) = 205.8, for O 2( g) = 205.2, for SO 2( g) = 248. Questions 47 are short free-response questions that require about 9 minutes each to answer and are worth 4 points each. Directions: Questions 13 are long free-response questions that require about 23 minutes each to answer and are worth 10 points each. Notice, however, that S o, unlike Δ H o f and Δ G o f for elements is not zero at 25 ☌įor example, using the data in an appendix for homonymic data, calculate the standard entropy changes, Δ S for the following reactions at 25☌:ĢH 2S( g) + 3O 2( g) → 2SO 2( g) + 2H 2O( l) YOU MAY USE YOUR CALCULATOR FOR THIS SECTION. 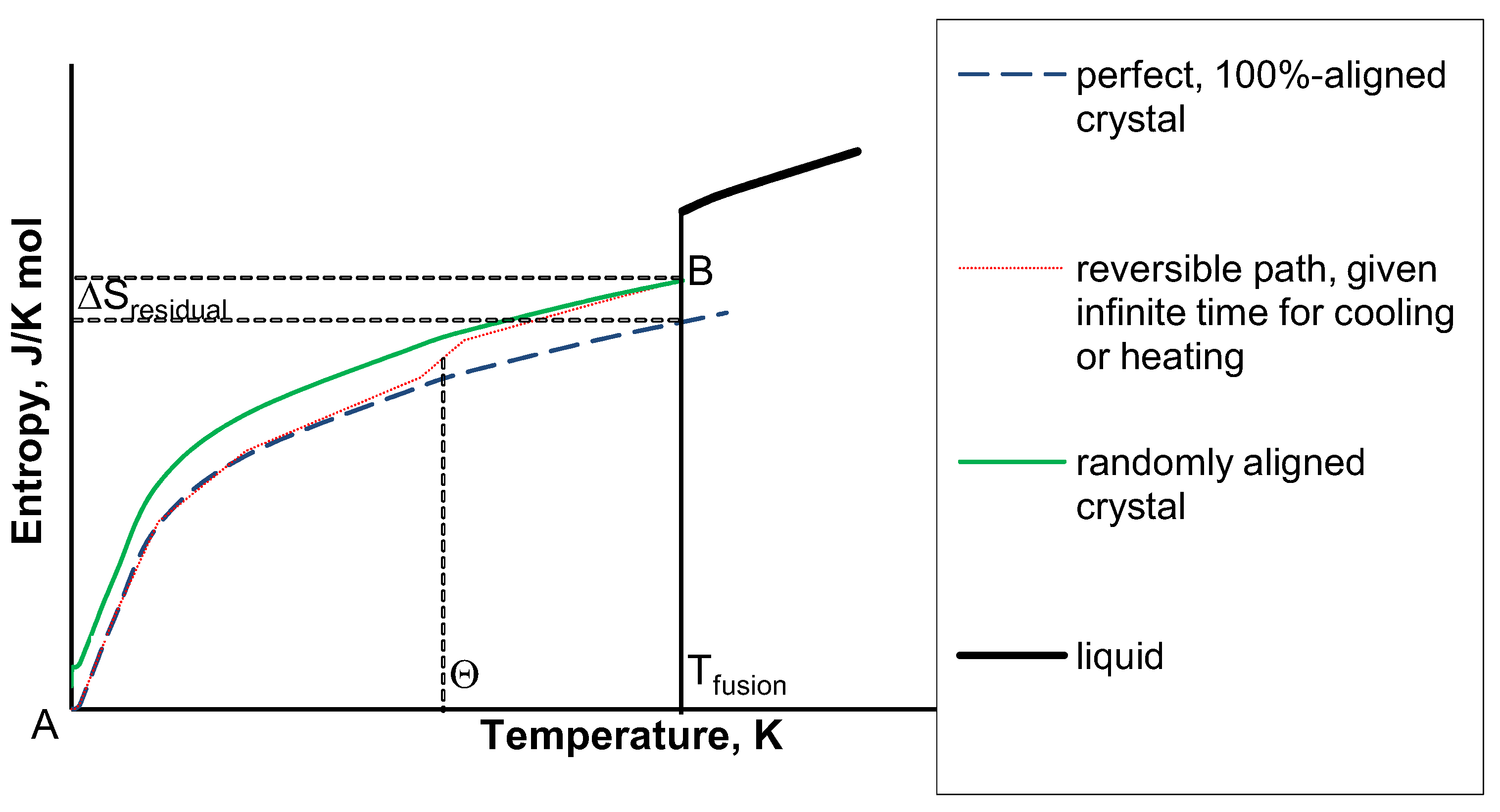
Solve the equation (specific heat at constant pressure Cp 4.1818 kJ/Kkg). Define final and initial temperature: Tf 20 C, Ti 100 C. You can also use this molarity calculator to find the mass concentration or molar mass. We will use the change in entropy formula: s Cp × ln (Tf / Ti), where Tf and Ti indicate the final and the initial temperature, respectively. Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. 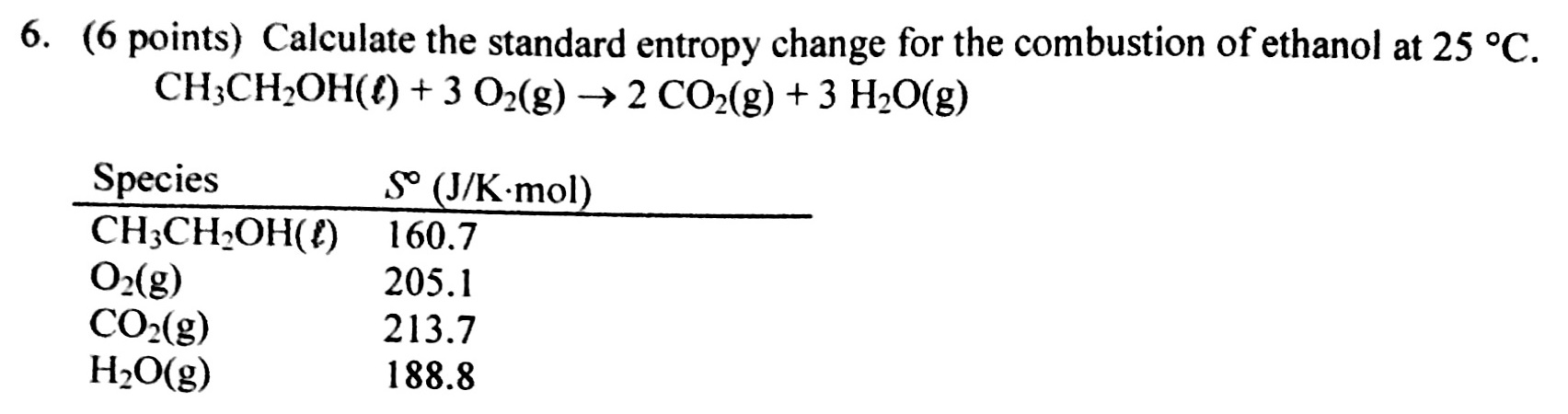
As mass / volume molarity × molar mass, then mass / (volume × molar mass) molarity. Earhart 2 of 2 All standard state, 25 ☌ and 1 bar (written to 1 decimal place). This is similar to how we calculate the enthalpy of a reaction from the standard enthalpies of formation. The term standard state is used to describe a reference state for substances, and is a help in thermodynamical calculations (as enthalpy, entropy and Gibbs. Convert the expressions above to obtain a molarity formula. ods, or phenomena: (a) absolute molar entropy. A positive value indicates an increase in entropy, while a negative value denotes a decrease in the entropy of a system. Usual units of standard molar entropy are joules per mole Kelvin (J/mol·K). The standard entropies of reactants and products are given in textbooks under the appendix for thermodynamics data. Use data from Appendix D to calculate the standard molar entropy change at 298.15 K for the conversion of. Standard molar entropy is defined as the entropy or degree of randomness of one mole of a sample under standard state conditions. (5.44) and the differential entropy (s) from Eq. The standard entropy change of the reaction is calculated by subtracting the standard entropies of the reactants multiplied by their stoichiometric coefficients from the standard entropies of the products multiplied by their stoichiometric coefficients: Using the absolute isotherms in Figure 1, the differential energy (u) was calculated from Eq. If S and H for a reaction have the same sign, then the sign of G depends on the relative magnitudes of the H and TS terms. S surr is the change in entropy of the surroundings. Regardless of the path the hikers have taken, they both are at the same altitude: The standard free-energy change can be calculated from the definition of free energy, if the standard enthalpy and entropy changes are known, using Equation 18.8.7: G H TS. The change in entropy of the surroundings after a chemical reaction at constant pressure and temperature can be expressed by the formula. Why would the number of neutrons in the nucleus a.  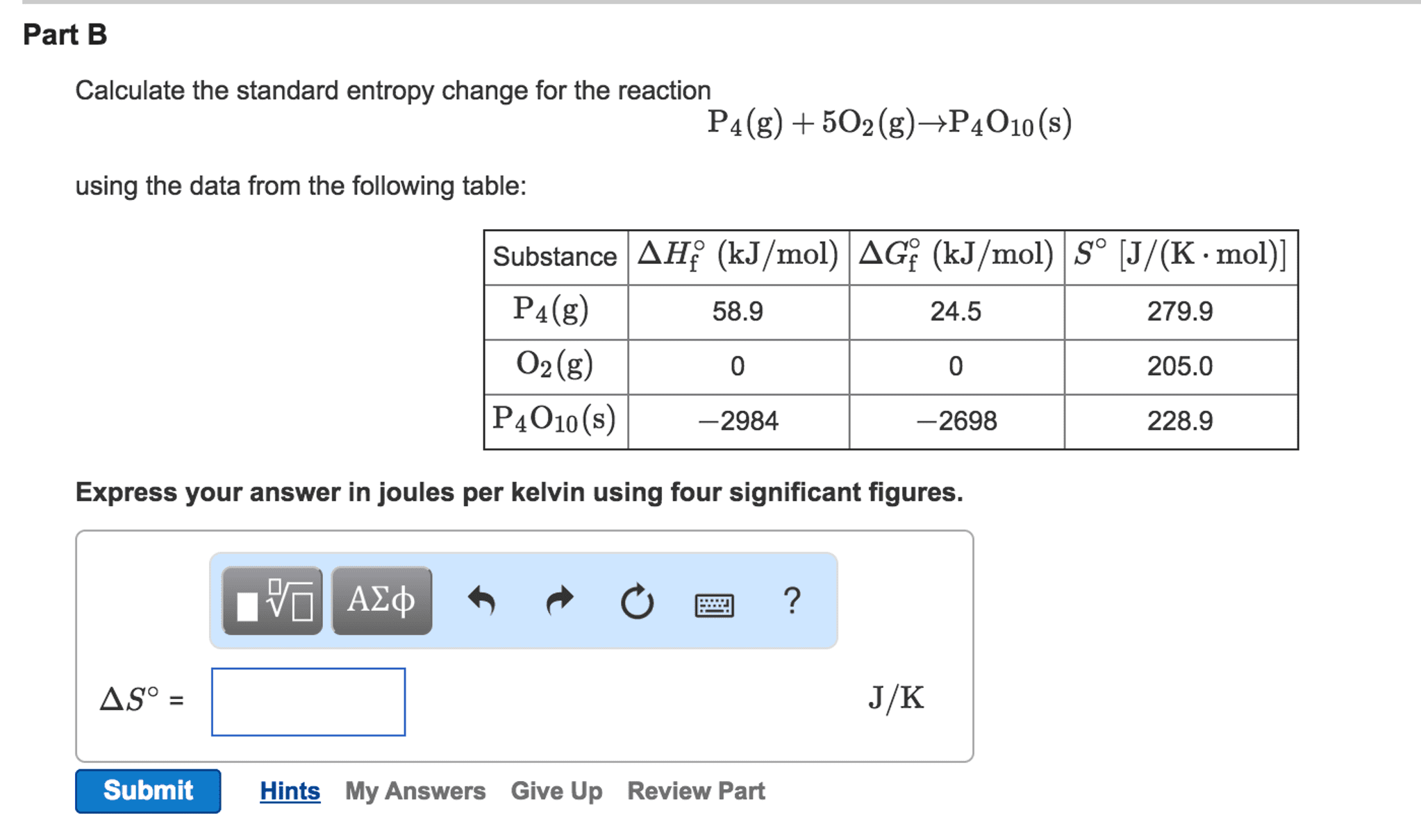
A classic example of a state function is attitude. Why is the entropy of deuterium more than that of hydrogen gas I thought about it but couldnt figure out a reason for why that would happen. Remember, a state function depends only on the initial and final values and not on the path that leads to these states. This term is used in condensed matter physics to describe the entropy at zero kelvin of a glass or plastic crystal referred to the crystal state, whose entropy is zero according to the third law of thermodynamics. be adopted for assigning absolute values to the enthalpy of various substances. Residual entropy is the difference in entropy between a non-equilibrium state and crystal state of a substance close to absolute zero. \hspace-.6pt\raise1.Because entropy is a state function, we can calculate the entropy change of a reaction if we know the standard entropies, S o of the reactants and products. energy Hm, molar entropy Sq, enthalpy change due to formation of substance. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
